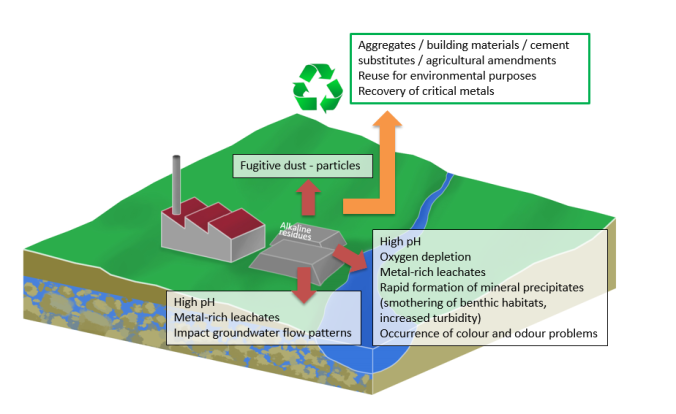
Over half a billion tonnes of alkaline (i.e. bleach-like) wastes are produced globally each year by carbon-intensive industries such as steel production, alumina refining and coal-fired power generation. Many attempts have been made to find re-uses for these residues such as in construction, but these are not sufficiently widespread to stop some of these wastes being disposed of in landfill sites. Here they require long term management as water that filters through the waste is toxic to aquatic life and dust generated as it is moved and stored is a public health hazard. It can take decades for these risks to fade. Recent studies have shown that these alkaline wastes contain large quantities of materials we would like to recover for re-use, particularly metals important to the green technologies of the future, such as vanadium, used in steel manufacture for offshore wind turbines, lithium and cobalt for vehicle fuel cells and rare earth elements crucial for next-generation solar power systems. The obvious solution: using the profits from recovering resources locked in the waste to pay for remediation of the pollution, is hampered by the environmental damage caused by digging up stored waste piles and the expense of extracting the metals from the waste using existing technology.
Our pilot tests have shown some of the environmental conditions under which these metals are preferentially dissolved from the wastes and into water passing through it. The metal solution then flows out of the bottom of the waste pile under gravity. The high concentrations mean that extracting metals from this solution becomes viable using existing technology that we propose to implement as part of this project. In effect the valuable resources are extracted without digging up the waste, which would cause environmental disturbance.
The resource recovery benefits are matched by benefits to the environment. Increased weathering of the wastes allows more CO2 to penetrate into the pile where it is locked away in significant quantities by reacting to form solid carbonate minerals. As elements like vanadium are pollutants as well as resources, recovery will eliminate the pollution alkaline waste weathering causes. Furthermore, the weathered waste piles have ideal conditions for nationally scarce, orchid-rich plant communities to become established, making them suitable for restoration to create habitat of high conservation value.
In order to turn the extremely promising results of our pilot studies into optimised, industry-ready processes we must better understand the specific mechanisms which control the metal release and develop a road map for negotiating the economic, legislative, environmental and societal challenges to the implementation of a new technology in an established industry with strict requirements for environmental protection. Our proposed research will tackle both these aspects in parallel. The combined package: recovery of the metal resources while suppressing dust, increasing carbon sequestration and treating the pollution caused, would be hugely beneficial to partners in our project from both industry (Tata Steel, Rio Tinto and the Minerals Industry Research Organisation) and environmental protection (Environment Agency).
The project will bring together key commercial partners with a multi-disciplinary team of environmental scientists, waste policy experts and specialists in systems analysis and stakeholder engagement to pave the way for transforming resource recovery and environmental remediation. This team will investigate the key obstacles to this transformation and identify potential remedies, such as lobbying for legislative change or making a clear business case for resource recovery.